FREMS-PDPN Participant Information Animation
The FREMS-PDPN Study is looking at whether a nerve stimulation device can reduce pain from the nerve damage diabetes can cause.
Peripheral Neuropathy happens because of nerve damage in the feet and legs.
This causes pain and discomfort that can badly affect sleep and quality of life.
The standard treatment for nerve pain is medication but it doesn’t work for everyone.
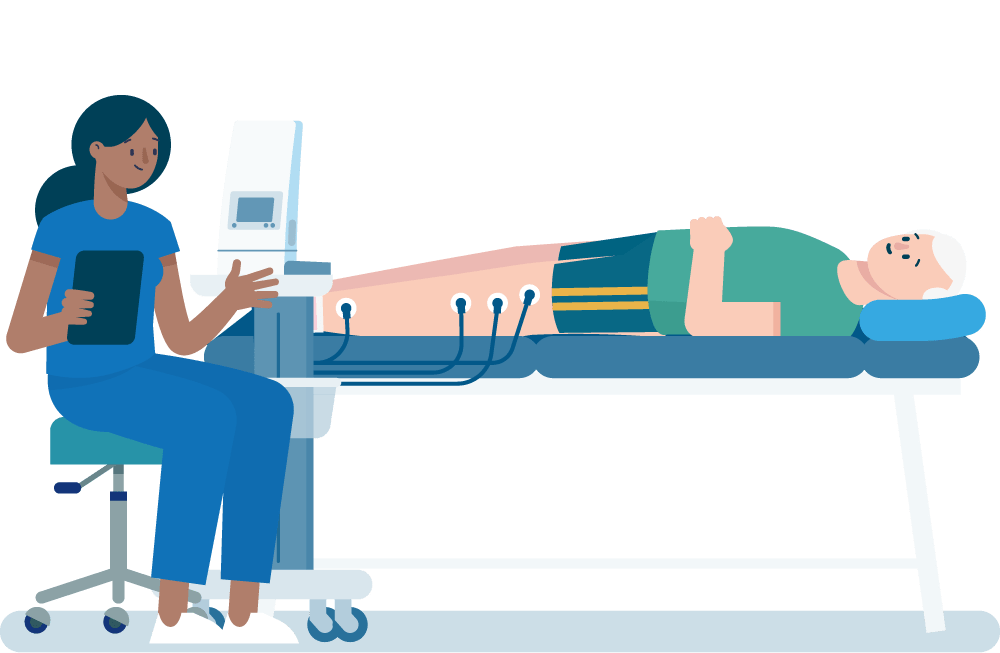
In this trial we are testing a treatment called FREMS.
FREMS is like TENS treatment, which is already used in the NHS for pain relief.
It uses the same kind of device, with pads stuck to the skin giving electrical pulses that stimulate the nerve ends.
Evidence suggests FREMS and TENS both work for pain in the short term.
And both are safe.
The difference is that FREMS delivers a different, changing electrical pulse.
We hope this trial will tell us if FREMS helps nerve pain better and for longer than TENS.
If you would like to take part we will screen you to see if the study is suitable for you.
First, you’ll need to give consent using an online form.
We will check your medical records.
We will ask you to complete a 7 day pain diary, sending us a daily score by text message.
Afterwards, if you are eligible we will arrange your first visit in person. At this visit you can ask any questions.
You can then take your time to decide what you would like to do.
If you decide to join the study...you will be asked to sign another online consent form.
Answer some questions about your medical history.
Complete questionnaires about your pain, sleep, mood, general health and wellbeing.
And we will do some tests.
If your doctor thinks the study is right for you, you will be randomly allocated to your treatment. Half to FREMS, half to TENS.
Neither you nor your doctor will know which you receive.
The rest of your care will continue as normal.
The treatment involves 10 sessions at hospital over two weeks, each about 40 minutes.
The electric pulse level is set by the nurse, based on how you feel.
Usually the electrical stimulation can be felt as a kind of buzzing, but it shouldn’t be painful.
We will ask you to send us your daily pain scores for 6 months.
In the final treatment session we will ask you to complete the questionnaire again.
This will be repeated in visits at three and six months after the treatment, where we will also check your progress and carry out a final medical.
Joining this study is completely voluntary and you can leave at any time.
Your decision will not affect the standard of care you receive.
The study team will be happy to answer any questions you may have.